Note: Descriptions are shown in the official language in which they were submitted.
CA 02480287 2004-09-22
WO 03/080599 PCT/IB03/01043
1
Stable Hydrate of a Muscarinic Receptor Antagonist
This invention relates to a stable solid hydrate of the muscarinic receptor
antagonist
(S)-2-~1-[2-(2,3-dihydrobenzofuran-5-yl)ethyl]-3-pyrrolidinyl}-2,2-
diphenylacetamide,
otherwise known as darifenacin (VII):
Ph
N ~~y~~ Ph
a
O NHZ
O
(VII)
In addition, the invention relates to pharmaceutical compositions containing
the
hydrate of the invention and to uses of said hydrate in medicine. Such
pharmaceutical
compositions are particularly relevant to the treatment of conditions for
which an
antagonist of muscarinic .receptors is required, such as irritable bowel
syndrome,
diverticular disease, oesophageal achalasia, chronic obstructive airways
disease, over
active bladder including symptoms of incontinence, urge and frequency, urinary
incontinence, neurogenic urinary urgency or pollakiuria, treatment of bladder
functional
disorder, urinary leakage, painful or difficult urination caused by neurogenic
bladder,
spastic or hypertonic bladder, dysfunctional bladder syndrome,
gastrointestinal
disorders including gastrointestinal hyperactivity, and relaxing effect on
intestinal
smooth muscle cells.
European patent 0388054 describes a family of 3-substituted pyrrolidine
derivatives
including darifenacin and pharmaceutically acceptable salts thereof as
muscarinic
receptor antagonists. The pharmaceutically acceptable salts include acid
addition
salts, specifically the hydrochloride, hydrobromide, hydrofluoride, sulphate
or
bisulphate, phosphate or hydrogen phosphate, acetate, besylate, citrate,
fumarate,
gluconate, lactate, maleate, mesylate, succinate and tartrate salts.
The hydrobromide salt of darifenacin has been the preferred compound for
medical
usage. The salt is produced from the corresponding anhydrous free base.
However, a
problem associated with the free base is that it is very unstable, having a
shelf life of
only one month. Additionally it can be difficult to produce the free base in a
sufficiently
pure form for pharmaceutical use.
CA 02480287 2004-09-22
WO 03/080599 PCT/IB03/01043
2
Surprisingly it has been found that this problem can be addressed by
synthesising the
hydrate of darifenacin for conversion to the hydrobromide salt rather than
employing
the free base to produce the hydrobromide salt. The solid hydrate has been
found to
remain stable for well over a year. Furthermore, it may be obtained to a level
of purity
suitable for pharmaceutical use. Conversion of the solid hydrate to the
medicinally
useful hydrobromide salt may be achieved via a facile transformation.
Accordingly, the present invention provides a stable solid hydrate of
darifenacin. It has
been shown by X-ray crystallography that the hydrate of the invention can be
isolated
as a compound possessing a stoichiometry of from 1:0.6 to 1:1 of
darifenacin:water.
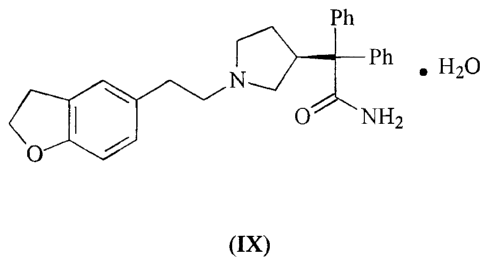
More particularly the invention provides a compound of the formula (IX):
Pn
N~~~B~Ph
.HBO
O NHZ
(IX)
In a preferred embodiment a compound of formula (IX) is characterised by an
infra-red
spectrum, run using single reflection ATR (attenuated total reflectance),
which shows
significant absorption bands at vmax. (crri'): 3625, 3516, 3440, 2948, 2806,
1699,
1622, 1597, 1578, 1488, 1471, 1445, 1378, 1353, 1325, 1312, 1280, 1242, 1196,
1152, 1119, 1102, 1086, 1024, 981, 939, 925, 900.
The compound of formula (IX) can also be characterised by a powder X-ray
diffraction
pattern obtained using copper radiation (~,= 0.15405 nm) which shows main
peaks at
8.39, 10.519, 13.272, 13.693, 15.908, 16.289, 16.855, 19.637, 21.135, 21.55,
21.722,
23.006, and 26.284 degrees 28.
It is still further characterised by its differential scanning calorimetry
(DSC) trace which
shows a sharp endotherm at 101 °C at a scan rate of 20°C/min.
CA 02480287 2004-09-22
WO 03/080599 PCT/IB03/01043
3
Infra Red spectroscopy was performed using a Nicolet Avatar 360 FT-IR
spectrometer.
Samples were run using single reflection ATR (attenuated total reflectance)
with the
spectrometer scanning in a spectral range of 650 cm' to 4000 cm'.
PXRD data were obtained using a SIEMENS D5000 powder X-ray diffractometer
fitted
with an automatic sample changer, a theta-theta goniometer, automatic beam
divergence slits, a secondary monochromator and a scintillation counter. The
samples
were prepared for analysis by packing the powder on to silicon wafer specimen
mounts. Each specimen was rotated whilst being irradiated with copper K-alphas
X-
rays (wavelength = 1.5406 angstroms) with the X-ray tube operated at
40kV/40mA.
The analyses were performed with the goniometer running in step-scan mode set
for a
5 second count per 0.02° step over a two theta range of 2° to
45°.
DSC was performed using a Perkin Elmer DSC-7 instrument fitted with an
automatic
sample changer. Approximately 3mg of sample was accurately weighed into a 50
microlitre aluminium pan and crimp sealed with a perforated lid. The samples
were
heated at 20°Clmin over the range 40°C to 250°C with a
nitrogen gas purge.
The invention further provides pharmaceutical compositions comprising a
hydrate of
the invention, as described above, together with a pharmaceutically acceptable
excipient, diluent or carrier.
Additionally, the invention provides the use of a hydrate of the invention, as
described
above" or of a pharmaceutical composition comprising a hydrate .of the
invention, as
described above" as a medicament.
Still further the invention provides the use of a hydrate of the invention, as
described
above" or of a pharmaceutical composition comprising a hydrate of the
invention, as
described above" for the manufacture of a medicament for curative or
prophylactic
treatment of a medical condition for which an antagonist of muscarinic
receptors is
indicated. Such conditions are irritable bowel syndrome, diverticular disease,
oesophageal achalasia, chronic obstructive airways disease, over active
bladder
(including symptoms of incontinence, urge and frequency), urinary
incontinence,
neurogenic urinary urgency or pollakiuria, treatment of bladder functional
disorder,
CA 02480287 2004-09-22
WO 03/080599 PCT/IB03/01043
4
urinary leakage, painful or difficult urination caused by neurogenic bladder,
spastic or
hypertonic bladder, dysfunctional bladder syndrome, gastrointestinal disorders
including gastrointestinal hyperactivity, and relaxing effect on intestinal
smooth muscle
cells.
Also provided by the invention is a method of treatment of a mammal to cure or
prevent a medical condition for which an antagonist of muscarinic receptors is
indicated, which comprises administering to said mammal an effective amount of
a
hydrate of the invention, as described above" or an effective amount of a
pharmaceutical composition comprising a hydrate of the invention, as described
above,.
The present invention also includes all suitable isotopic variations of a
hydrate of the
invention, as described above,. An isotopic variation of a hydrate of the
invention, as
described above, is defined as one in which at least one atom is replaced by
an atom
having the same atomic number but an atomic mass different from the atomic
mass
usually found in nature. Examples of isotopes that can be incorporated into a
hydrate
of the invention, as described above, include isotopes of hydrogen, carbon,
nitrogen
and oxygen such as 2H, 3H, '3C, '4C, 'SN, "Q and '$O respectively. Certain
isotopic
variations of a hydrate of the invention, as described above" for example,
those in
which a radioactive isotope such as 3H or'4C is incorporafied, are useful in
drug and/or
substrate tissue distribution studies. Tritiated, i.e. 3H, and carbon-14, i.e.
'4C, isotopes
are particularly preferred for their ease of preparation and detectability.
Further,
substitution with isotopes such as deuterium, i.e. 2H, may afford certain
therapeutic
advantages resulting from greater metabolic stability, for example, increased
in vivo
half-life or reduced dosage requirements and hence may be preferred in some
circumstances. Isotopic variations of a hydrate of the invention, as described
above,
can generally be prepared by conventional procedures such as by the
illustrative
methods or by the preparations described in the examples and preparations
hereafter
using appropriate isotopic variations of suitable reagents.
Hydrates of the invention, as described above, can be administered alone but
will
generally be administered in admixture with a suitable pharmaceutical
excipient, diluent
or carrier selected with regard to the intended route of administration and
standard
CA 02480287 2004-09-22
WO 03/080599 PCT/IB03/01043
pharmaceutical practice. For example, a hydrate of the invention, as described
above,
can be administered orally, buccally or sublingually in the form of tablets,
capsules,
multi-particuiates, gels, films, ovules, elixirs, solutions or suspensions,
which may
contain flavouring or colouring agents, for immediate-, delayed-, modified-,
sustained-,
5 pulsed-, or controlled-release applications. Hydrates of the invention, as
described
above, may also be administered as fast-dispersing or fast-dissolving dosage
forms or
in the form of a high energy dispersion or as coated particles. Suitable
formulations of
a hydrate of the invention, as described above, may be in coated or uncoated
form as
desired.
Such solid pharmaceutical compositions, for example tablets, may contain
excipients
such a microcrystalline cellulose, lactose, sodium citrate, calcium carbonate,
dibasic
calcium phosphate and glycine, disintegrants such as starch (preferably Born,
potato or
tapioca starch), sodium starch glycollate, croscarmellose sodium and certain
complex
silicates, and granulation binders such as polyvinylpyrrolidone,
hydroxypropylmethylcellulose (HPMC), hydroxypropylcellulose (HPC), sucrose,
gelatin
and acacia. Additionally, lubricating agents such as magnesium stearate,
stearic acid,
glyceryl behenate and talc may be included.
Solid compositions of a similar type may also be employed as fillers in
gelatin or HPMC
capsules. Preferred excipienfs in this regard include lactose, starch, a
cellulose, milk
sugar or high molecular weight polyethylene glycols. For aqueous suspensions
and/or
elixirs, a hydrate of the invention, as described above, may be combined with
various
sweetening or flavouring agents, colouring matter or dyes, with emulsifying
andlor
~5 suspending agents and with diluents such as water, ethanol, propylene
glycol and
glycerin, and combinations thereof.
Hydrates of the invention, as described above, can also be administered
parentera!!y,
for example, intravenously, intra-arterially, intraperitoneally,
intrathecally,
intraventriculariy, intraurethrally, intrasternally, intracranially,
intramuscularly or
subcutaneously, or they may be administered by infusion or needleless
injection
techniques. For such parenteral administration they are best used in the form
of a
sterile aqueous solution which may contain other substances, for example,
enough
salts of glucose to make the solution isotonic with blood. The aqueous
solutions
CA 02480287 2004-09-22
WO 03/080599 PCT/IB03/01043
6
should be suitably buffered (preferably to a pH of from 3 to 9), if necessary.
The
preparation of suitable parenteral formulations under sterile conditions is
readily
accomplished by standard pharmaceutical techniques well-known to those skilled
in
the art.
For oral and parenteral administration to human patients, the daily dosage
level of the
hydrates of the invention, as described above, will usually be from 1.5 to 30
mg (in
single or divided doses). The physician in any event will determine the actual
dosage
which will be most suitable for any individual patient and it will vary with
the age, weight
and response of the particular patient. The above dosages are exemplary of the
average case. There can, of course, be individual instances where higher or
lower
dosage ranges are merited and such are within the scope of this invention.
Hydrates of the invention, as described above, can also be administered
intranasally or
by inhalation and are conveniently delivered in the form of a dry powder
inhaler or an
aerosol spray presentation from a pressurised container, pump, spray, atomiser
or
nebuliser, with or without the use of a suitable propellant, e.g.
dichlorodifluoromethane,
trichlorofluoromethane, dichlorotetrafluoroethane, a hydrofluoroalkane such as
1,1,1,2-
tetrafluoroethane (HFA 134ATM) or 1,1,1,2,3,3,3-heptafluoropropane (HFA
227EATnn),
carbon dioxide or other suitable gas. In the case of a pressurised aerosol,
the dosage
unit may be determined by providing a valve to deliver a metered amount. The
pressurised container, pump, spray, atomiser or nebuliser may contain a
solution or
suspension of the active compound, e.g. using a mixture of ethanol and the
propellant
as the solvent, which may additionally contain a lubricant, e.g. sorbitan
trioleate.
Capsules and cartridges (made, for example, from gelatin) for use in an
inhaler or
insufflator may be formulated to contain a powder mix of a hydrate of the
invention, as
described above, and a suitable powder base such as lactose or starch.
Aerosol or dry powder formulations are preferably arranged so that each
metered dose
or "puff' contains from 0.2 mg to 3.0 mg of a hydrate of the invention, as
described
above, for delivery to the patient. The overall daily dose with an aerosol
will be in the
range of from 0.5 mg to 10.0 mg of a hydrate of the invention, as described
above,
which may be administered in a single dose or, more usually, in divided doses
throughout the day.
CA 02480287 2004-09-22
WO 03/080599 PCT/IB03/01043
7
Alternatively, a hydrate of the invention, as described above, can be
administered in
the form of a suppository or pessary, or it may be applied topically in the
form of a gel,
hydrogel, lotion, solution, cream, ointment or dusting powder. Hydrates of the
invention, as described above, may also be dermally or transdermally
administered, for
example, by the use of a skin patch. They may also be administered by the
pulmonary
or rectal routes.
Alternatively, hydrates of the invention, as described above, may be
administered topically to the skin, mucosa, dermally or transdermally, for
example, in
the form of a gel, hydrogel, lotion, solution, cream, ointment, dusting
powder, dressing,
foam, film, skin patch (for example, but not limited to, the following types,
reservoir,
matrix, drug-in-adhesive, multilaminate polymer sysytem), wafers, implant,
sponges,
fibres, bandage, microemulsions and combinations thereof. For such
applications, a
hydrate of the invention, as described above, can be suspended or dissolved
in, for
example, a mixture with one or more of the following: mineral oil, liquid
petrolatum,
white petrolatum, propylene glycol, polyoxyethylene polyoxypropylene compound,
emulsifying wax, glycerine, silicone fluids, fixed oils, including synthetic
mono- or
diglycerides, and fatty acids and fatty acid esters, including oleic acid,
water, sorbitan
monostearate, a polyethylene glycol, liquid paraffin, polysorbate 60, cetyl
esters wax,
cetearyl alcohol, 2-octyldodecanol, benzyl alcohol, alcohols such as ethanol.
Alternatively, penetration enhancers may be used, for example but not limited
to the
following in the Journal of Pharm. Sciences, October 1999 by Finnin and Morgan
"Transdermal Penetration Engancers: Applications, Limitations and Potential".
The
following may also be used polymers, carbohydrates, proteins, phospolipids in
the
form of nanoparticles (such as niosomes or liposomes) or suspended or
dissolved. In
addition, they may be delivered using iontophoresis, electroporation,
phonophoresis,
sonophoresis and needle free injections.
Hydrates of the invention, as described above, may also be used in combination
with a
cyclodextrin. Cyclodextrins are known to form inclusion and non-inclusion
complexes
with drug molecules. Formation of a drug-cyclodextrin complex may modify the
solubility, dissolution rate, bioavailabilty and/or stability property of a
drug molecule.
Drug-cyclodextrin complexes are generally useful for most dosage forms and
administration routes. As an alternative to direct complexation with the drug
the
CA 02480287 2004-09-22
WO 03/080599 PCT/IB03/01043
cyclodextrin may be used as an auxiliary additive, e.g, as a carrier, diluent
or
solubiliser. Alpha-, beta- and gamma-cyclodextrins are most commonly used and
suitable examples are described in WO-A-91111172! WO-A-94/02518 and WO-A-
98/55148.
The compounds of the invenfiion may be prepared as shown below:
Ph Ph Ph
Ph
RCN 'CN
phenol
N aq. HBr ~ ~ .HBr ~ P~h
N,N-carbonyldiimidazole, N~ph
Ts (II) * ethyl acetate, ~ ~/ JcN
(I) toluene O ~ / O
OH
(I~
O / O
sodium borohydride,
(III) boron tritluoride-
tetrahydrofuran
complex,
toluene, tetrahydrofuran,
piperazine,
propau-2-of
Ph Ph
N~Ph aqueous hydrobromic acid, \ N~Ph
.HBr cN methanol ~ c~N
O / O
M> (~
potassium hydroxide,
2-medtylbutan-2-of
potassium hydroxide,
2-methylbutan-2-of
r Ph
N~\\s~Ph
/ O~NHz
O
(VII)
(i) amberlife resin, solvent
(ii) toluene
Ph Ph
N~~~Ph aqueous hydrobromic acid, ~ N Ph
O ..II~~NHx butan-2-one O~ HBr O NH,
Toluene
(VIII)
(
B ~ acetonitrile
water
Ph r Ph
N~~'~Ph aqueous hydrobromic acid, ~ N~~~Ph
HZQ O NH2 butan-2-one O~ HBr O NH2
(I~ (
Scheme 1.
CA 02480287 2004-09-22
WO 03/080599 PCT/IB03/01043
9
Surprisingly it has been found that darifenacin hydrate may be obtained in a
pharmaceutically pure form from a solution of darifenacin which is subjected
to a resin
treatment and then converted to the hydrate via a toluene solvate (see steps A
and B
in Scheme 1.). Darifenacin toluene solvate may be directly converted to the
hydrobromide, however this conversion does not allow for flexibility in the
production
plant scheduling because the toluene solvate is not stable over medium to long
term
storage periods. This additions( burden on the manufacturing process may be
overcome by converting darifenacin toluene solvate to darifenacin hydrate,
which is
stable over long periods, and so conversion to darifenacin hydrobromide can
then be
performed when required without fear that in the meantime compound (IX) will
have
degraded in quality.
Accordingly, the present invention further provides a process for providing a
hydrate of
the invention, as described above, in pharmaceutically pure form by subjecting
darifenacin to a resin treatment followed by conversion to a toluene solvate
which is in
turn converted to said hydrate. A solution of darifenacin in a suitable
organic solvent
or aqueous organic solvent mixture is combined with the resin and the
resulting
mixture is stirred at between ambient temperature and reflux. Subsequently the
darifenacin solution is separated from the resin by filtration. Preferably the
resin is a
quaternary ammonium hydroxide resin. The resin treatment may be pertormed in
batch mode or in a continuous processing mode. The hydrate may be further
elaborated to give an acid addition salt of darifenacin. Preferably, the acid
addition salt
is the hydrobromide salt.
Further, the present invention provides a novel intermediate for the provision
of a
hydrate of the invention, as described above, in the form of the toluene
solvate of
darifenacin. It is envisaged that other solvates of darifenacin, for example
the ethyl
acetate solvate, may be employed in the place of the toluene solvate.
It has been shown by X-ray crystallography that compound (VIII) possesses a
1:1
stoichiometry, i.e. one molecule of darifenacin and one molecule of toluene in
an
asymmetric unit.
CA 02480287 2004-09-22
WO 03/080599 PCT/IB03/01043
Compound of formula (VIII) is characterised by an infra-red spectrum, run
using single
reflection ATR (attenuated total reflectance), which shows significant
absorption bands
at v,,,ax. (Cm ~ ): 3463, 3342, 3299, 3285, 3022, 2925, 2825, 1673, 1614,
1490, 1440,
1384, 1333, 1319, 1243, 1195, 1152, 1130, 1115, 1102, 1028, 1003, 980, 939,
926,
5 907.
This compound can also be characterised by a powder X-ray diffraction pattern
obtained using copper radiation (~.= 0.15405 nm) which shows main peaks at
12.572,
12.754, 15.978, 17.419, 18.537, 18.889, 20.78, 21.562, 22.437, 22.736, 23.767,
10 24.075, 24.266, 25.35, 25.762, 27.214, and 29.716 degrees 28.
It is still further characterised by its differential scanning calorimetry
(DSC) trace which
shows a sharp endotherm at 92°C at a scan rate of 20°Clmin.
The following examples illustrate the preparation of compounds disclosed in
Scheme
1.:-
Example 1:
(S)-2,2-diphenyl-2-(3-pyrroiidinyl)acetonitrile hydrobromide (II)
Ph
Ph
'CN
H .HBr
A mixture of (S)-2,2-diphenyl-2-(1-tosyl-3-pyrrolidinyl)acetonitrile (I) [see
European
Patent Publication No. 0388054] (83.8 Kg, 201.2 moles), 48% aqueous
hydrobromic
acid (419 L, 5 L/Kg of compound I) and phenol (16.8 Kg, 0.2 Kg/Kg of compound
I) is
heated at reflux for 3 hours. The mixture is cooled and extracted with
dichloromethane
(1 x 560 Kg, 1 x 523 Kg). The extracts are combined and washed with aqueous
sodium chloride solution (15 Kg in 150 Kg of water). The organic layer is
concentrated
and essentially replaced with ethyl acetate to a total volume of about 440 L.
Hexane
(276 Kg) is added at 40°C and product is collected at 0-5°C by
filtration. The (S)-2,2-
CA 02480287 2004-09-22
WO 03/080599 PCT/IB03/01043
11
diphenyl-2-(3-pyrrolidinyl)acetonitrile hydrobromide is washed with chilled
ethyl acetate
and dried under vacuum at 60°C. Yield 52.8 Kg (76%).
v = 3441, 2940, 2745, 2455, 2246, 1972, 1886, 1806, 1596, 1585, 1561, 1494,
1450,
1392, 1289, 1255, 1217, 1159, 1104, 1070, 1034, 1002, 967, 917, 899, 833, 766,
750,
702, 664, 645, 546, 496, 472 crri'.
'H NMR (300 MHz, CDCI3): 8 = 2.12 (2H, m), 3.15 (1 H, m), 2.96 (3H, m), 3.76
(1 H,
quin, J 8 Hz), 7.25-7.41 (6H, m), 7.47 (4H, t, J 8 Hz), 9.23 (1 H, br, s),
9.43 (1 H, br).
LRMS (electrospray, positive ion): m/z [MH+] 263.
Optical rotation: [a] 365 = -55.9°
Example 2:
(S)-3-(cyanodiphenylmethyl)-1-[2-(2,3-dihydrobenzofuran-5-
yl)acetyl]pyrrolidine (IV)
Ph
N~\~Ph
RCN
O~ O
To a cooled (0-5°C) slurry of 2-(2,3-dihydrobenzofuran-5-yl)acetic acid
(III) (9.85 Kg,
55.3 moles) in ethyl acetate (115 L) is added carbonyldiimidazole (8.97 Kg,
55.3
moles). The reaction is stirred at 5-10°C for 1 hour prior to the
addition of (S)-2,2-
diphenyl-2-(3-pyrrolidinyl)acetonitrile hydrobromide (II) (17.25 Kg, 50.2
moles). The
reaction is allowed to warm up to 20-25°C and stirred for an additional
3 hours. The
reaction mixture is washed with 2N aqueous hydrochloric acid (42 L) then
aqueous
sodium bicarbonate (2.1 Kg in 42 L of water). The ethyl acetate solution is
concentrated and essentially replaced with toluene to furnish a solution of
product in
toluene with a total ° volume of about 43 L. The assumed yield of (S)-3-
(cyanodiphenylmethyl)-1-[2-(2,3-dihydrobenzofuran-5-yl)acetyl]pyrrolidine is
100%
(21.2 Kg) and is employed directly in the preparation of compound V.
CA 02480287 2004-09-22
WO 03/080599 PCT/IB03/01043
12
v = 3448, 3059, 3026, 2973, 2948, 2878, 2236, 1959, 1890, 1811, 1719, 1643,
1600,
1491, 1449, 1421, 1362, 1336, 1297, 1241, 1219, 1198, 1159, 1125, 1102, 1034,
1002, 983, 944, 917, 892, 836, 804, 764, 752, 701, 667, 646, 618, 576, 550,
469, 424,
405 cni'.
For this compound, two structural conformations exist giving rise to 'doubled-
up'
signals for some of the resonances. 'H NMR (300 MHz, CDCi3): 8 = 1.85-2.20
(2H,
m), 3.16 & 3.18 (2H, t, J 9 Hz), 3.20-3.85 (7H, m), 4.54 & 4.55 (2H, t, J 9
Hz), 6.68 &
6.70 (1 H, d, J 9 Hz), 6.83 & 6.94 (1 H, d, J 9 Hz), 7.05 & 7.12 (1 H, s),
7.22-7.48 (1 OH,
m).
LRMS (electrospray, positive ion): m/z [MH+] 423.
Optical rotation: [a] 365 = +85.9°
Example 3:
(S)-2-{1-[2-(2,3-dihydrobenzofuran-5-yl)ethyl]-3-pyrrolidinyl}-2,2-
diphenylacetonitrile (V)
Ph
N~\~ Ph
CN
O /
To a cooled (0°C) mixture of (S)-3-(cyanodiphenylmethyl)-1-[2-(2,3-
dihydrobenzofuran-
5-yl)acetyl]pyrrolidine (IV) as a toluene solution (7.43 Kg active, 17.59
moles) and
sodium borohydride (0.87 Kg, 23 moles) in tetrahydrofuran (29.7 L) is added
boron
trifluoride tetrahydrofuran complex (4.31 Kg, 30.81 moles) at such a rate as
to maintain
the temperature of the reaction below 10°C. The reaction is warmed to
ambient
temperature and stirred for a further 4 hours. Aqueous piperazine solution is
added
and the mixture is heated at reflux for 8 hours. The aqueous layer is
separated and
washed with 1 % aqueous sodium chloride solution (22.3 L) at 40°C. The
organic layer
is concentrated and essentially replaced by isopropyl alcohol at atmospheric
pressure
to a total volume of about 30 L. The product crystallises on cooling and is
collected at
0-5°C by filtration. (S)-2-{1-[2-(2,3-dihydrobenzofuran-5-yl)ethyl]-3-
pyrrolidinyl}-2,2-
CA 02480287 2004-09-22
WO 03/080599 PCT/IB03/01043
13
diphenylacetonitrile (V) is washed with chilled isopropyl alcohol and dried
under
vacuum at 50°C. Yield 6.34 Kg (88%).
v = 3441, 3088, 3056, 3032, 2947, 2924, 2884, 2856, 2790, 2744, 2237, 1955,
1883,
1809, 1614, 1596, 1489, 1448, 1385, 1353, 1338, 1322, 1290, 1245, 1216, 1195,
1148, 1130, 1101, 1076, 1033, 1016, 1003, 980, 944, 921, 891, 847, 819, 799,
764,
750, 701, 674, 658, 646, 573, 563, 540, 504, 491, 427, 403 cm-' .
'H NMR (300 MHz, CDCI3); 1.86 (1 H, m), 2.10 (1 H, m), 2.38 (1 H, t, J 9 Hz),
2.52 (1 H,
q, J 8 Hz), 2.59-2.75 (4H, m), 2.84 (1 H, m), 3.02 (1 H, dt, J 4 & 9 Hz), 3.16
(2H, t, J 9
Hz), 3.47 (1 H, m), 4.53 (2H, t, J 9 Hz), 6.67 (1 H, d, J 8 Hz), 6.90 (1 H, d,
J 8 Hz), 7.00
(1 H, s), 7.23-7.40, (6H, m), 7.46 (4H, t, J 8 Hz).
LRMS (eiectrospray, positive ion): m/z [MH+] 409.
Optical rotation: [a] 365 -- +31.8°
Example 4:
(S)-2-{1-[2-(2,3-dihydrobenzofuran-5-yl)ethyl]-3-pyrrolidinyl}-2,2-
diphenylacetonitrile
hydrobromide (VI)
Ph
N~\~Ph
~f ~U
CN .HBr
O
To a slurry of (S)-2-(1-[2-(2,3-dihydrobenzofuran-5-yl)ethyl]-3-pyrrolidinyl}-
2,2-
diphenylacetonitrile (V) (30.0 g, 0.073 moles) in methanol (150 mL) is added
48%
aqueous hydrobromic acid (13.6 g, 0.081 moles) maintaining the temperature
below
40°C. The mixture is heated at reflux for 1 hour. The batch is cooled
to 0°C, and the
product is collected by filtration, washed with methanol (60 mL) and dried at
50°C
under vacuum to give (S)-2-{1-[2-(2,3-dihydrobenzofuran-5-yl)ethyl]-3-
pyrrolidinylj-2,2-
diphenylacetonitrile hydrobromide (VI) (33.5 g, 93%).
CA 02480287 2004-09-22
WO 03/080599 PCT/IB03/01043
14
v = 3440, 3059, 3002, 2931, 2893, 2856, 2653, 2624, 2548, 2496, 2471, 2239,
1960,
1888, 1812, 1615, 1599, 1493, 1450, 1394, 1363, 1332, 1294, 1242, 1159, 1129,
1106, 1088, 1073, 1035, 1003, 981, 941, 889, 830, 766, 751, 725, 703, 666,
645, 582,
548, 534, 500, 476, 423 cm-'.
'H NMR (300 MHz, CDCI3); 2.08 {1 H, m), 2.46 (1 H, m), 2.75 (1 H, q, J 10 Hz),
2.69-
3.33 (7H, m), 3.70 (1 H, m), 3.83 (1 H, m), 4.09 (1 H, m), 4.54 (2H, t, J 9
Hz), 6.69 {1 H,
d, J 8 Hz), 6.92 (1 H, d, J 8 Hz), 7.06 (1 H, s), 7.27-7.50 (1 OH, m), 12.08
(1 H, br).
LRMS (electrospray, positive ion): m/z [MHt] 409.
Optical rotation: [a] 365 = +90.0°
Example 5:
(S)-2-{1-[2-(2,3-dihydrobenzofuran-5-yl)ethyl]-3-pyrrolidinyl}-2,2-
diphenylacetamide
toluene solvate (VIII)
Ph
N ~~~~~ Ph
O NHZ
o Toluene
Method 1: A slurry of potassium hydroxide (48.7 g, 0.87 moles) in 2-
methylbutan-2-of
(175 mL) is heated at 50-60°C. After 1 hour (S)-2-{1-[2-(2,3-
dihydrobenzofuran-5-
yl)ethyl]-3-pyrrolidinyl}-2,2-diphenylacetonitrile hydrobromide (VI) (25.0 g,
0.051 moles)
is added and the resulting mixture is heated at reflex for 20 hours. The
reaction
mixture is cooled to ambient temperature and water (125 mL) is added
maintaining the
temperature below 30°C. The mixture is stirred for 15 minutes, then
allowed to settle
and the organic phase is separated. The organic phase is washed with aqueous
sodium chloride (125 mL of 5%wlw solution) to provide a solution of (S)-2-{1-
[2-(2,3-
dihydrobenzofuran-5-yl)ethyl]-3-pyrrolidinyl}-2,2-diphenylacetamide as a
solution in 2-
methylbutan-2-of (VII). The solution is heated at reflex in the presence of
Amberlite~
resin (37.5 g) for 22 hours then cooled to ambient temperature. The Amberlite~
resin
CA 02480287 2004-09-22
WO 03/080599 PCT/IB03/01043
is removed by filtration and washed with 2-methylbutan-2-of (25 mL). The
combined 2-
methylbutan-2-of phases are concentrated and essentially replaced by toluene
to a
final volume of approximately 140 mL. The toluene solution is cooled to
0°C during
which time crystallisation occurs. (S)-2-~1-[2-(2,3-dihydrobenzofuran-5-
yl)ethyl]-3-
5 pyrrolidinyl}-2,2-diphenylacetamide toluene solvate (VIII) is collected by
filtration,
washed with chilled toluene (25 mL) and dried at 35°C under vacuum.
Yield (22.2 g,
84%).
Method 2: A slurry of potassium hydroxide (40 g, 0.71 moles) in 2-methylbutan-
2-of
10 (140 mL) is heated at 50-60°C. After 1 hour, (S)-2-~1-[2-(2,3-
dihydrobenzofuran-5-
yl)ethyl]-3-pyrrolidinyl}-2,2-diphenylacetonitrile (V) (20 g, 0.049 moles) is
added and
the resulting mixture is heated at reflex for approximately 20 hours. The
reaction
mixture is cooled and water (100 mL) is added maintaining the temperature
below
34°C. The mixture is stirred for 30 minutes and the organic phase is
separated. The
15 organic phase is washed with aqueous sodium chloride (100 mL of 5%w/w
solution) to
provide a solution of the product as a solution in 2-methylbutan-2-ol. The
solution is ,
heated at reflex in the presence of Amberlite0 resin (30 g) for 9 hours then
cooled to
ambient temperature. The Amberlite0 resin is removed by filtration and washed
with
2-methylbutan-2-of (20 mL). The combined 2-methylbutan-2-of phases are
concentrated and essentially replaced by toluene to a final volume of
approximately 80
mL. The toluene solution is cooled to 0°C during which time
crystallisation occurs. (S)-
2-{1-j2-(2,3-dihydrobenzofuran-5-yl)ethyl]-3-pyrrolidinyl}-2,2-
diphenylacetamide toluene
solvate (VIII) is collected by filtration, washed with toluene (70 mL) and
dried at 35°C
under vacuum. Yield (17.2 g, 68%).
v = 3463, 3342, 3299, 3285, 3022, 2925, 2825, 1673, 1614, 1490, 1440, 1384,
1333,
1319, 1243, 1195, 1152, 1130, 1115, 1102, 1028, 1003, 980, 939, 926, 907
crri'.
'H NMR (300 MHz, d6-DMSO): 8 = 1.57 (1 H, m), 1.93 (2H, m), 2.3-2.5 (6H, m),
2,82
(1 H, t, J 9), 3.11 (2H, t, J 9), 3.62 (1 H, m), 4.47 (2H, t, J 9), 6.62 (1 H,
d, J 8), 6.82 (1 H,
d, J 8), 6.99 (1 H, s), 7.08 (2H, m), 7.2-7.4 (10H, m). Signals were observed
for
toluene corresponding to a molar ratio of 1 at 2.3 and are present underneath
the
CA 02480287 2004-09-22
WO 03/080599 PCT/IB03/01043
16
aromatic region for (S)-2-{1-[2-(2,3-dihydrobenzofuran-5-yl)ethyl]-3-
pyrrolidinyl}-2,2-
diphenylacetamide.
Optical rotation: [a] 365 = -119.0°
Example 6:
(S)-2-{1-[2-(2,3-dihydrobenzofuran-5-yl)ethyl]-3-pyrrolid inyl}-2,2-d
iphenylacetamide
hydrate (IX)
Ph
N~~.,/~Ph
\ ~/ .H20
O NHz
O
A solution of (S)-2-(1-[2-(2,3-dihydrobenzofuran-5-yl)ethyl]-3-pyrrolidinyl}-
2,2-
diphenylacetamide toluene solvate (VIII) (16 g, 0.031 moles) in acetonitrile
(320 mL) is
concentrated under reduced pressure at ambient temperature. The resulting foam
is
dissolved in acetonitrile (48 mL) to which is added water (1~:1~ mL) dropwise
at ambient
temperature. The solution is stirred at ambient temperature until
crystallisation occurs
and is allowed to stir overnight. The (S)-2-{1-[2-(2,3-dihydrobenzofuran-5-
yl)ethyl]-3-
pyrrolidinyl}-2,2-diphenylacetamide hydrate (IX) is collected by filtration
and dried
under vacuum at ambient temperature. Yield (10.4 g, 76%).
v = 3625, 3516, 3440, 2948, 2806, 1699, 1622, 1597, 1578, 1488, 1471, 1445,
1378,
1353, 1325, 1312, 1280, 1242, 1196, 1152, 1119, 1102, 1086, 1024, 981, 939,
925,
900 crri'.
'H NMR (300 MHz, ds-DMSO): 8 = 1.57 (1 H, m), 1.93 (2H, m), 2.3-2.5 (6H, m),
2,82
(1 H, t, J 9), 3.11 (2H, t, J 9), 3.62 (1 H, m), 4.46 (2H, t, J 9), 6.62 (1 H,
d, J 8), 6.81 (1 H,
d, J 8), 6.99 (1 H, s), 7.07 (2H, m), 7.2-7.4 (10H, m).
Water content by Karl Fischer: 2.7% w/w
Optical rotation: [a] 365 - -120.7°
CA 02480287 2004-09-22
WO 03/080599 PCT/IB03/01043
PC22018
17
Example 7:
(S)-2-{1-[2-(2,3-dihydrobenzofuran-5-yl)ethyl]-3-pyrrolidinyl}-2,2-
diphenylacetamide
hydrobromide (X)
Ph
N~~y~~Ph
.HBr
O NHz
°
Method 1: A solution of (S)-2-{1-[2-(2,3-dihydrobenzofuran-5-yl)ethylj-3-
pyrrolidinyi}-
2,2-diphenylacetamide toluene solvate (VIII) (30.4 g, 0.059 moles) in butan-2-
one (213
mL) is warmed to 33 °C to attain solution and then cooled to 15
°C. 48% aqueous
hydrobromic acid(9.9 g, 0.059 moles) is then added and the mixture stirred at
15 °C for
1 hour and 0 °C for 2 hours. The (S)-2-{1-[2-(2,3-dihydrobenzofuran-5-
yl)ethyl]-3
pyrrolidinyl}-2,2-diphenylacetamide hydrobromide (X) is collected by
filtration, washed
' with butan-2-one (65 mL) and dried under vacuum at 50 °C for 18
hours. Yield
(24.6 g, 83%).
Method 2: To a solution of (S)-2-{1-[2-(2,3-dihydrobenzofuran-5-yl)ethyl]-3-
pyrrolidinyl}-2,2-diphenylacetamide hydrate (IX) (3.60 g, 0.0081 moles) in
butan-2-one
(30 mL) is added 48% aqueous hydrobromic acid(1.36 g, 0.0081 moles). The
mixture
is stirred at 20 °C for 1 hour and 0 °C for 1 hours and the (S)-
2-{1-[2-(2,3-
dihydrobenzofuran-5-yl)ethyl]-3-pyrrolidinyl}-2,2-diphenylacetamide
hydrobromide (X)
is collected by filtration, washed with butan-2-one (10 mL) and dried under
vacuum at
50 °C for 18 hours. Yield (3.90 g, 95%). m.p. = 232°C.
v = 3468, 3211, 3052, 2995, 2870, 2693, 2586, 1668, 1585, 1492, 1442, 1243,
983,
850 cm'.
'H NMR (300 MHz, CDCI3): 8 = 2.10-2.23 (1 H, m); 2.81-2.99 (2H, m); 3.00-3.15
(4H, m); 3.15 (2H, t); 3.18-3.29 (1 H, m); 3.48 (1 H, t); 3.69 (1 H, s); 3.80-
3.95
(1 H, m); 4.52 (2H, t); 5.58 (1 H, bs); 5.62 (1 H, bs); 6.63 (1 H, d); 6.84 (1
H, d);
7.01 (1 H, s); 7.19-7.40 (10H, m); 11.48 (1 H, bs).
Optical rotation: [a,] 589 = + 46.0°